|
11/25/2023 0 Comments Sequential screening vs maternit21
Currently, all marketed DNA-based prenatal screening tests are proprietary, available through the tests’ manufacturers, although some manufacturers are entering into marketing agreements with certain reference laboratories. 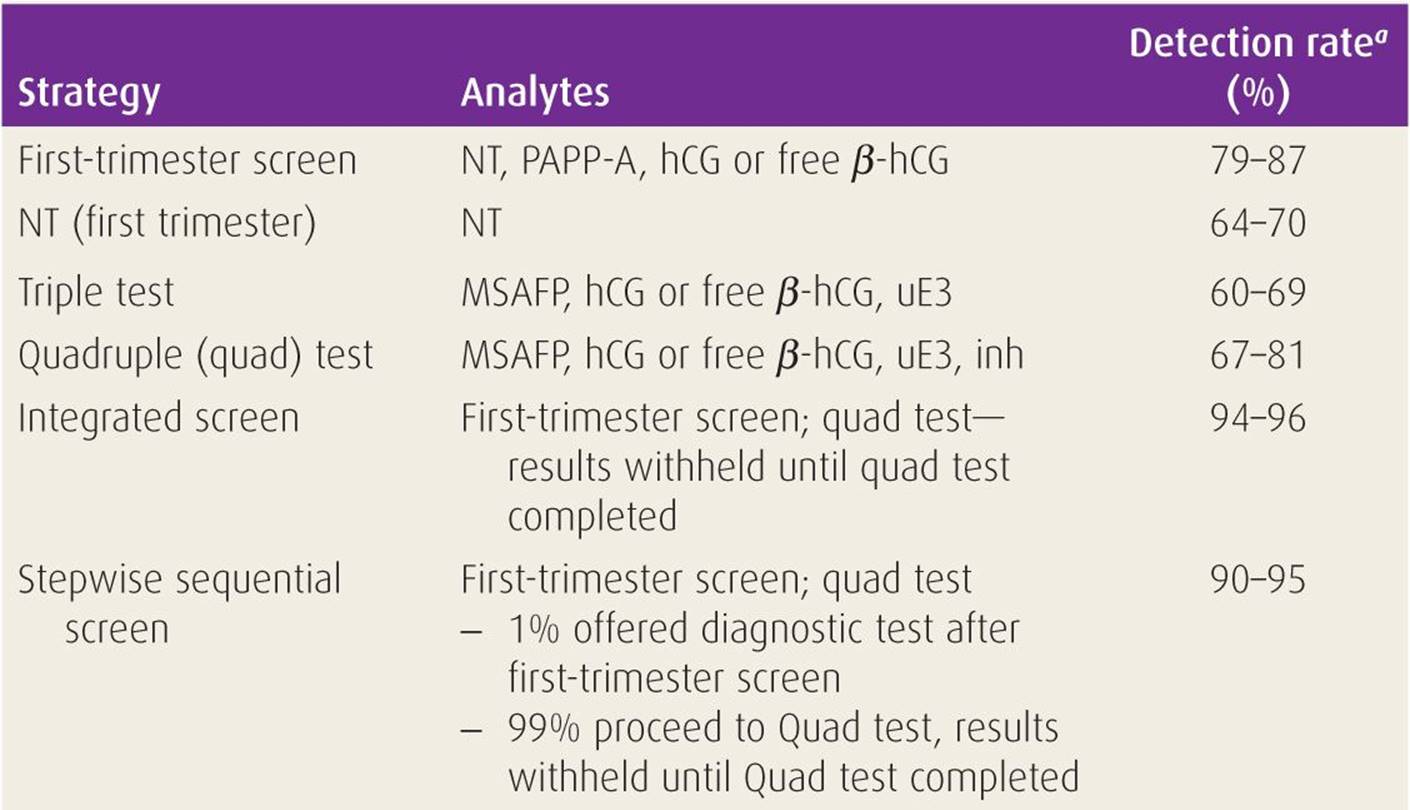
The fourth method (Panorama™, Natera), uses genotyping by assessment of single nucleotide polymorphisms (SNPs) in maternal and fetal DNA in maternal circulation. Three of the current tests are based on counting methods that compare expected quantities of genetic material from a given chromosome to observed quantities in maternal serum, and then calculate the likelihood that deviations from expected quantitative ratios are due to fetal aneuploidy. Two of the tests currently on the market (MaterniT21™ by Sequenom and Verifi™ by Verinata) use massive parallel (“next generation”) sequencing of random DNA fragments in maternal serum, and the other two tests (Harmony™ by Ariosa and Panorama™ by Natera) involve more targeted DNA amplification methods. Each is in the form of a proprietary laboratory developed test (LDT) each established, marketed, and performed by the test’s manufacturer. Currently there are four companies offering DNA-based maternal plasma prenatal screening (often referred to as “non-invasive prenatal testing” or NIPT). 
Late 2011 saw the first commercial release of maternal serum prenatal aneuploidy screening methods based on analysis of maternal serum free DNA, rather than assessment of biochemical analytes. However, the best performing biochemical / NT-based prenatal screens still have detection rates only in the 90 – 95% range, false positive rates in the 1 to 2% range, and positive predictive values of around 5%. Maternal serum biochemical and NT screening is a useful and cost-effective method for calculating age-adjusted risk for the presence of NTD and fetal aneuploidy, and as an aid in determining which women should undertake the risk of undergoing definitive diagnostic procedures such as chorionic villus sampling or amniocentesis. Regardless of the screening panel used, the objective is to derive likelihood ratios by which to adjust the age-associated risk of carrying an aneuploidy pregnancy. įetal nuchal translucency (NT) measurement by certified ultrasonographers is also used in first trimester, sequential, and full integrated screens. Since then, additional maternal serum biochemical markers, including human chorionic gonadotropin (HCG), unconjugated estriol (uE3), dimeric inhibin A (DIA), and pregnancy-associated plasma protein-A (PAPP-A) have expanded prenatal screening from second trimester “triple” and “quad” screening (AFP, uE3, HCG, DIA) to first trimester screening (HCG and PAPP-A), to sequential and integrated first and second trimester screening. In the decade following the description of increased maternal serum alpha fetoprotein (MSAFP) levels in women carrying fetuses affected by open neural tube defects (NTDs), an association was reported between low levels of MSAFP and fetal aneuploidy, most notably Down syndrome (trisomy 21).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
